Introduction: Secondary cytopenias occurring after hematopoietic cell transplant (HCT) are an observed complication of graft-versus-host disease (GVHD). Causes include peripheral immune mediated destruction (hemolytic anemia or thrombocytopenia), usually associated with autoantibody production, and peripheral destruction associated with microangiopathy (TMA). However, cases have been observed where marrow production of the affected lineage is abnormal. Immune-mediated cases are treated with increased immunosuppression, e.g., steroids or calcineurin inhibitors (CNI), and TMA with complement inhibition. To better understand the mechanisms and treatment of hypoproductive cytopenias in GVHD, we combined analyses of hematopoietic stem and progenitor cells (HSPC) and peripheral blood and marrow activated T cells. We describe 3 cases of GVHD-associated cytopenias marked by HSPC depletion and presence of CD25 (IL-2R alpha subunit)-positive activated T cells in the marrow that responded to combinations of immunosuppression containing basiliximab (basil) anti-CD25 monoclonal antibody blockade.
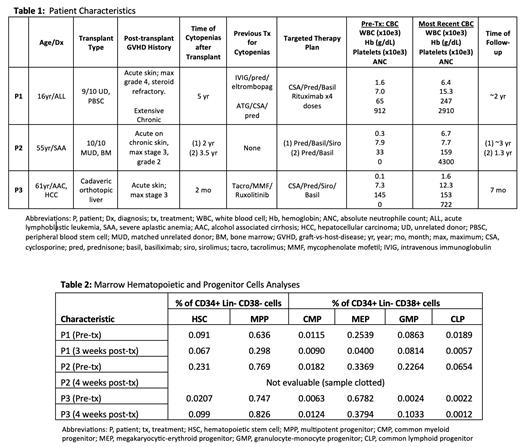
Methods/Results: We describe combined HSPC and T cell analyses in 3 patients (P) with severe GVHD-associated cytopenias. P1 was at 5 years post-HCT for acute lymphoblastic leukemia, P2 at 2 years post-HCT for severe aplastic anemia (SAA), and P3 at 1.5 months post-liver transplant for alcohol use disorder-associated cirrhosis and hepatocellular carcinoma ( Table 1). Because of refractory cytopenias, all were being considered for HCT. However, marrow analyses revealed that each had normal frequencies of phenotypic HSC, with variable decreases in cellularity and frequencies of committed progenitors ( Table 2). Chimerism analyses showed that 100% of CD34+ cells in P1 and P2 were of donor origin, and in P3 of host origin. All 3 marrows had activated CD3+ T cells per expression of CD69 and/or CD38, and frequency of CD25 on >10% of T cells. In addition, P1 had evidence of IgG bound to committed progenitor populations. The treatment strategy was to block IL-2 signaling with basil (induction of 20 mg on day 1, 4, 7, 14, 21, 28, followed by alternate week and then monthly dosing for 2-9 months), combined with suppression of IL-2 production with steroid and/or CNI. After induction, repeat analyses of the blood and marrow T-cells confirmed complete blockade of CD25. Therapy was also individualized for specific clinical conditions or as noted on marrow evaluation. P1 received prednisone, CNI, and rituximab for a presumed anti-HSPC autoantibody. At 9 months he was weaned off all therapy and remains on no immunosuppressive treatment 2 years later. P2 did not receive CNI due to chronic renal failure but received steroids and later sirolimus. At ~18 months, his cytopenias recurred, but subsequently responded to basil re-induction and steroids. P3 has received prednisone, CNI, and sirolimus indefinitely for prevention of liver rejection and/or hepatocellular carcinoma relapse. P1 had no viral reactivation; P2 had CMV pneumonia with his second course responsive to antiviral therapy, completion of basil, and tapering of steroids; and P3 had CMV reactivation responsive to antiviral therapy and EBV viremia. P3 has a recent diagnosis of EBV+ intrahepatic B cell lymphoma. In all cases, blood counts normalized except P2 who has anemia due to CKD.
Discussion: HSPC analyses in these patients with GVHD and cytopenias demonstrate that peripheral changes are not due to loss of HSC, but instead to quantitative and/or qualitative defects in committed progenitor populations, likely mediated by activated CD25+ T cells. In addition, the presence of IgG on HSPCs in P1 demonstrates a novel role for antibody-mediated humoral suppression in some cases. The presence of HSC indicates that there may be no need for re-transplant. Previous studies of SAA have shown activated T cells to be drivers of aplasia and is the basis for immune suppressive therapy (anti-thymocyte globulin and CNI). Our analyses of patients with GVHD-associated cytopenias demonstrates that targeting of IL-2 signaling via combined CD25 receptor blockade and pharmacologic inhibition of IL-2 production can correct the marrow defects and cytopenias, albeit with need for attention to viral infections. Targeting CD25 in patients with evidence of CD25+ activated T cells is a rational approach to the difficult problem of GVHD-associated marrow suppression and cytopenias.
OffLabel Disclosure:
John:vertex: Membership on an entity's Board of Directors or advisory committees; bluebird bio: Consultancy, Membership on an entity's Board of Directors or advisory committees. Kwon:Jasper Therapeutics: Current Employment, Current equity holder in publicly-traded company. Shizuru:Jasper Therapeutics: Consultancy, Current equity holder in publicly-traded company.
Basiliximab is not formally FDA approved for use in GVHD though is commonly used off label regardless of CD25 expression.